A 45-year-old woman with a suspected malignant right pleural effusion undergoes video-assisted thoracoscopic surgery for drainage of the effusion and pleural biopsies. During the course of the procedure, the patient suffers a cardiac arrest, but she is successfully resuscitated following the Advance Cardiovascular Life Support (ACLS) protocol. Postoperatively, she is critically ill and intubated in the intensive care unit (ICU). During her initial ICU course, she suffers from respiratory failure, acute renal failure, "shock liver" (hepatic ischemia), and a right upper extremity deep venous thrombosis. Her past medical history is only significant for hypertension, and she has no history of smoking or drinking. She has no known drug allergies. On postoperative day 3, the plastic surgery service is consulted with regard to a change in the upper extremity examination.
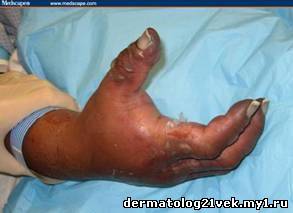
The patient is seen in the ICU, where she is noted to be critically ill, and on both norepinephrine and phenylephrine to support her blood pressure. With this support, her blood pressure is 137/71 mm Hg, and her heart rate is 134 bpm. She is on independent 2-lung ventilatory support for her severe respiratory failure. Her temperature is 97.2°F (36.2°C). On physical examination, her right upper extremity distal to the elbow is noted to be extremely swollen and tense. The skin of the forearm and dorsal hand is blistered, and some of the blisters are unroofed and weeping. The distal hand and fingers are darkly discolored and cool, and, although the brachial pulse is present, the radial and ulnar pulses are not palpable. Attempts to passively flex and extend the fingers and wrist are inhibited by the tense nature of the soft tissues. The fingers are all in a slightly flexed position, and the nail beds are blackened.
Numerous laboratory studies had been previously sent by the ICU team. The complete blood cell (CBC) count reveals a white blood cell (WBC) count of 24.9 ? 103/µL (24.9 ? 109/L), hemoglobin of 11.9 g/dL (119 g/L), hematocrit of 36.2% (0.362), and a platelet count of 40 ? 103/µL (40 ? 109/L). Her creatinine is 2.7 mg/dL (238.68 µmol/L). The arterial blood gas results are a pH of 7.47, pO2 of 119 mm Hg, pCO2 of 33 mm Hg, and HCO3 of 25 mEq/liter.
Top of Form
Questions answered incorrectly will be highlighted.
What condition had led to the current findings of this patient's right upper extremity examination?
Hint: Look closely at the position of the hand.
Cellulitis
Necrotizing fasciitis
Arterial insufficiency
Compartment syndrome
What condition had led to the current findings of this patient's right upper extremity examination?
Hint: Look closely at the position of the hand.
Your Colleagues Responded:
Cellulitis
2%
Necrotizing fasciitis
17%
Arterial insufficiency
18%
Compartment syndrome
Correct Answer
61%
Discussion:
This is a prime example of compartment syndrome of the right forearm and hand. The clinical examination of the patient's arm was highly suggestive of compartment syndrome. A 21-gauge needle was connected to a central venous pressure monitoring device and was passed into the forearm and hand in the tense areas. Elevated compartment pressure readings of 56 mm Hg in the hand and 32 mm Hg in the forearm were documented. The etiology of the development of compartment syndrome in this patient was believed to be the result of the deep venous thrombosis in the affected extremity. Although laboratory studies are useful in the overall care of the critically ill patient, they are not absolutely necessary to confirm this particular diagnosis.
Compartment syndrome is an increase in tissue pressure within a closed space that, if left untreated, can compromise circulation and lead to ischemia. It can occur in any area of the body in which a compartment is present. These include the hand, forearm, upper arm, lower extremity, abdomen, and buttock. The distal lower extremity is the most commonly affected location.[1]
There are numerous etiologies for compartment syndrome, the most common being trauma with a bony or crush injury.[1] Other sources of increased compartment fluid and pressure include bleeding, anticoagulation therapy, electrical injury, infection, reperfusion injury, envenomation, extremely vigorous exercise, or intravenous (IV) infiltration.[1,3] Other causes of decreased compartment space and increased pressure include constrictive dressings, thermal injuries, or tight fascial closures.[1,2,3] Lying on a limb, such as can occur in drug overdoses, intoxication, or other neurologic events, can also cause acute compartment syndrome.[1,3] The clinician should be on the lookout for this syndrome if a history of any of the above is present. Deep venous thrombosis, the suspected underlying cause for the development of compartment syndrome in this case, is a relatively rare etiology. The index of suspicion should be raised if there is severe pain in addition to a suggestive history.
The diagnosis of compartment syndrome is mainly clinical. A thorough history taking that addresses the above etiologies will be helpful; however, in patients who are unable to communicate, the diagnosis must be made based solely on physical signs. Pain out of proportion to tenderness on examination is the classic physical finding; other findings include muscle weakness, swelling, firmness of the compartment itself, and paresthesias. Accentuation of the pain by passive stretching of the muscles is an extremely reliable indicator of compartment syndrome. This test can be performed to evaluate any of the 3 compartments in the forearm or the 10 compartments in the hand, and it can then be used to guide the subsequent treatment accordingly.[3]
On inspection, the ischemic forearm is generally held in supination, with the wrist and interphalangeal joint in flexion and the metacarpophalangeal joint in extension (see Figure 1).[1] It is important to note that arterial pressure is generally higher than compartmental pressure, and the distal pulses may be intact even with ongoing tissue compromise. The loss of pulses is a late sign.[1,2,3] Direct measurement of compartment pressures can also be undertaken, but the results should not exclude the diagnosis of compartment syndrome if strong clinical suspicion is present. This measurement is taken with a 21-gauge needle attached to a central venous pressure monitor (or other similar measuring device). There is a wide range of pressures that is reported to be the threshold for performing fasciotomy, with 30 mm Hg being a widely quoted value.[1] Regardless, if there is any clinical suspicion for compartment syndrome, or any elevated compartment pressures, serial examinations should be undertaken, and an experienced surgeon should evaluate the patient.
Although laboratory studies are commonly ordered in these patients, they are not absolutely crucial to establishing the diagnosis. These studies may include a CBC count, complete metabolic profile, prothrombin time (PT), partial thromboplastin time (PTT), creatine phosphokinase (CPK), and urinalysis. An initial urinalysis can be compared with subsequent studies to assess for the presence of urine myoglobin and rhabdomyolysis. Radiographs of the affected extremity are also often ordered, but these studies are likewise not essential.
If compartment syndrome is left untreated, progressive muscle ischemia ensues. The approximate upper limit of muscle viability is 6 hours following an ischemic insult. A Volkmann ischemic contracture may be the end result of an unrecognized or untreated compartment syndrome. This is described as the fingers in flexion and the wrist in flexion and pronation.[3] The outcome can be catastrophic, with possible complete loss of the limb or, in rare situations, loss of life. Once the diagnosis is made, the treatment is an emergency decompressive fasciotomy. There is little role for conservative treatment in compartment syndrome. General anesthesia is the preferred method for an emergent fasciotomy. As described in the literature, a volar fasciotomy is performed, with a standard carpal tunnel incision that is carried to the ulnar aspect of the wrist. This should avoid injury to the radial artery, radial nerve, and median nerve, and further allow for a radial-based skin flap to facilitate subsequent closure.
From this position, the incision is carried in a curvilinear fashion toward the central and proximal aspect of the forearm. The superficial flexor carpi ulnaris, flexor carpi radialis, and palmaris longus muscles are all released. The deeper flexor pollicis longus, flexor digitorum superficialis, and flexor digitorum profundus muscles are all released, as needed. If the dorsal compartments are still tense after the volar fasciotomy, then incisions can be made longitudinally on the dorsal forearm to allow release. There are 10 compartments in the hand, and all may need release. The dorsal compartments can be released by longitudinal incisions on the dorsum of the hand, and the thenar and hypothenar muscle groups can be decompressed through small longitudinal incisions. After decompression, hemostasis is achieved and the muscles are inspected. Obviously, necrotic muscle should be debrided, but questionable muscle should be left and evaluated again later. A moist, sterile dressing should be applied to the open wounds. The hand is then splinted in the neutral position. Most patients have their wounds primarily closed in 7-14 days, and skin grafting can be performed if primary closure is not feasible. The skin should not be closed if there remains any question of muscle viability. Scarring after this procedure is often severe, and patients may be given the option of scar revision in the future, if they so desire.[1,2,3]
In this patient, aggressive fluid resuscitation was continued in order to decrease the effects of the acute renal failure and rhabdomyolysis. Because of the hemodynamic instability of the patient and the risk of transport to the operating room, general anesthesia was administered in the ICU by the anesthesia staff. A volar forearm fasciotomy was performed through a carpal tunnel-type incision, which was carried toward the ulnar aspect of the wrist and then curved centrally toward the proximal forearm. Upon incision, the forearm muscles appeared pink and healthy, and upon release, the dorsal forearm also became noticeably softer. Then, 4 linear incisions were placed on the dorsum of the hand to allow for release of the dorsal compartments. The wounds were left open, and moist dressing changes were undertaken. The hand was splinted in the neutral position.
References
Zamboni WA, Kiraly EM. Compartment Syndrome. In: Achauer BM, Eriksson E, Guyuron B, et al. Plastic Surgery Indications, Operations, and Outcomes. Vol. 4. Philadelphia, PA: Elsevier, 2000.
Mathes SJ, Hentz VR. Plastic Surgery. Volume 7. 2nd ed. Philadelphia, PA: Elsevier; 2006: 600-602.
Gulgonen, A. Compartment Syndrome. In: Green D, Hotchkiss R, Pederson WC, Wolfe SW. Green’s Operative Hand Surgery. 5th Ed. Philadelphia, PA: Elsevier, 2005.
CME Test
Top of Form
Questions answered incorrectly will be highlighted.
Which of the following physical findings indicative of compartment syndrome is typically the last to occur?
Pain
Loss of pulses
Paresthesias
Pressure
Paralysis of the extremity
What is the optimal treatment once a diagnosis of acute compartment syndrome is confirmed?
Elevation of the extremity
Ice
Initiation of broad-spectrum antibiotics
Decompressive fasciotomy
Splinting of the extremity
Bottom of Form
Which of the following physical findings indicative of compartment syndrome is typically the last to occur?
Your Colleagues Responded:
Pain
3%
Loss of pulses
Correct Answer
56%
Paresthesias
3%
Pressure
1%
Paralysis of the extremity
35%
What is the optimal treatment once a diagnosis of acute compartment syndrome is confirmed?
Your Colleagues Responded:
Elevation of the extremity
1%
Ice
0%
Initiation of broad-spectrum antibiotics
1%
Decompressive fasciotomy
Correct Answer
96%
Splinting of the extremity
 Главная
Главная  A 45-year-old woman with a suspected... (см. перевод ниже) - Форум
A 45-year-old woman with a suspected... (см. перевод ниже) - Форум Регистрация
Регистрация Вход
Вход
