Whitish Bullae on the Fingers: What is the Diagnosis? Ana Nogueira MD; Ana F. Duarte MD; Paulo Morais MD; Sofia Magina MD; Filomena Azevedo MD
Abstract
A 56-year-old woman presented with a 6-month history of bullae and whitish plaques of the fingers. She had chronic renal failure, but no joint pains or evidence of arthritis. An aspirate of fluid from a bulla revealed uric acid crystals and serum uric acid was elevated at 16.2 mg/dL. A diagnosis of tophaceous gout was made.
Case Synopsis
A 56-year-old obese woman, admitted for acute arterial ischemia of the lower limb that required revascularization, presented with whitish plaques and bullae of the fingers that evolved over a six month period.
Although she suffered from chronic renal insufficiency and atrial fibrillation, she was not taking any regular medication. She was an alcoholic and did not seek regular medical attention. She displayed painful soft whitish-yellow bullous plaques with an erythematous rim, affecting mainly the finger pads (Figures 1 & 2). She had no signs of arthritis. On punch biopsy a whitish fluid was extruded, which was observed microscopically (Figure 3) and showed uric acid crystals. Her uric acid level was 16.2 mg/dL (normal up to 6.1).
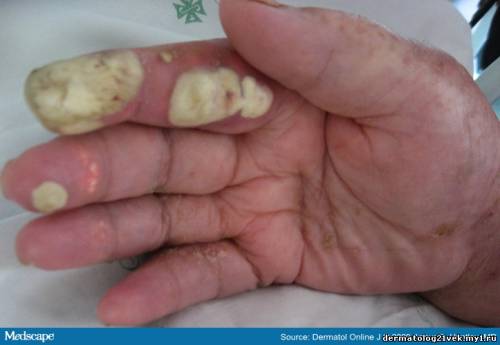
Figure 1.
Whitish-yellow soft bullous plaques on finger pads
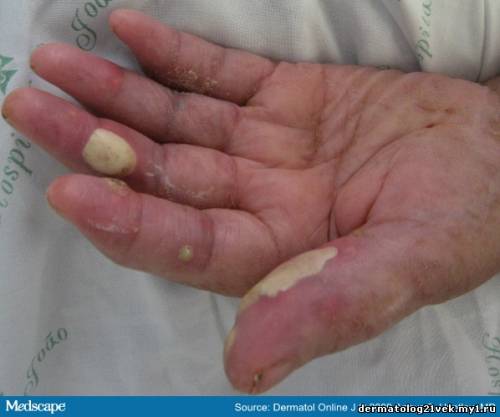
Figure 2.
Whitish-yellow soft bullous plaques on finger pads

Figure 3.
Fine needle aspiration cytology from a finger pad (×100)
Discussion
Gout is a metabolic disease caused by a disturbance in purine metabolism in which crystals of monosodium urate from supersaturated extracellular fluids are deposited in tissues, such as joints, kidneys, and soft tissues, producing an inflammatory response. It usually presents with recurrent painful monoarthritis, mainly in the lower extremities.[1-3] Without treatment, tophi emerge about 10 years after the onset of the disease and rarely, as in our case, can come up without previous gouty arthritis.[2,4] They appear as quite firm white to yellowish papules or nodules with white pasty content and have a predilection for the distal interphalangeal joints, olecranon bursa, dorsal aspect of the proximal interphalangeal and metacarpophalangeal joints, and dorsal toes.[1] Finger pad lesions, as this patient exhibited, are unusual and arise mostly in post-menopausal women without previous evidence of gout.[2,5] Nevertheless, tophi can develop almost anywhere. As they progressively enlarge, spontaneous drainage may take place through a skin opening or by rupture and ulceration of bullous lesions.[1]
The prevalence of gout is closely related to wealth and is highest in industrialized countries. Major risk factors include high purine consumption, alcohol intake, use of loop and thiazide diuretics, obesity, hypertension, and renal dysfunction. Other situations that increase the risk of hyperuricemia include psoriasis, myeloproliferative and lymphoproliferative disorders, dehydration, postoperative state, metabolic acidosis, sarcoidosis, chronic lead intoxication, hyperparathyroidism, ketosis, polycystic kidney disease and certain enzyme defects. Medications also favor this condition, such as low-dose aspirin, cyclosporine, ethambutol, pyrazinamide, ritonavir, levodopa, and nicotinic acid.[6] The risk of gout increases with increasing hyperuricemia, although serum uric acid levels may be elevated without clinical evidence of gout.[3] This patient displayed some risk factors because of her obesity, alcoholism, and chronic renal disease. Also, because she was post-menopausal, the odds of developing gout was increased because of deprivation of the protective effects of estrogen on uric acid levels. Post-operative state also probably aggravated her hyperuricemia.
The diagnosis of gout is considered when clinical, laboratory, and imaging findings are consistent with the disease. Serum uric acid levels are not very reliable; they can be normal even in an acute attack and there is a wide prevalence of asymptomatic hyperuricemia. The presence of tophi strongly supports the diagnosis. The disclosure of monosodium urate crystals in synovial fluid, although not always feasible, is confirmatory. Radiographic findings are only reliable later in the disease, in the chronic tophaceus stage.[1,3]
The differential diagnosis of tophaceous gout includes calcium pyrophosphate deposition disease (pseudogout), calcinosis cutis, and rheumatoid or cholesterol nodules.[1,3,4,6]
The treatment of chronic tophaceous gout involves identifying and eliminating correctable risk factors as well as starting urate-lowering therapy with allopurinol (a xanthine oxidase inhibitor, first choice) or probenecid (an uricosuric agent, second choice). Generally, a uric acid level of 6 mg/dL is targeted. When initiating these drugs, concomitant low-dose colchicine may be used for three to six months to lessen the risk of flare-ups. Other drugs such as febuxostat and rasburicase are being tried although still not routinely used.[3] Our patient unfortunately did not initiate any of these therapies because she developed a urinary tract infection that evolved to multiorgan dysfunction, septic shock, and ultimately caused her death before any therapeutic intervention for her gout was attempted.
 Главная
Главная  Белесоватые пузыри на пальцах: каков диагноз? - Форум
Белесоватые пузыри на пальцах: каков диагноз? - Форум Регистрация
Регистрация Вход
Вход
